

The United States became the first country to surpass 10 million confirmed cases of COVID-19 on Monday, the milestone comes as experts warn of a surge this fall and winter.
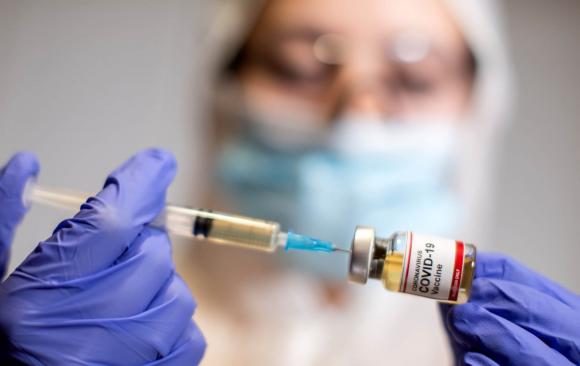
But, at the same time, Pfizer announced Monday morning that late-stage clinical trial data show that its vaccine candidate is over 90 percent effective in preventing COVID-19.
Governor Andrew Cuomo sees great promise in today's news that Pfizer had developed a vaccine. "The good news is the Pfizer tests look good and we'll have a vaccine shortly, the bad news is it's about two months before Joe Biden takes over and that means this administration will be implementing a vaccine plan." Cuomo said. Over the weekend, Governor Andrew M. Cuomo also updated New Yorkers on the state's progress during the ongoing COVID-19 pandemic.
"Cases are increasing across the country and the globe -- and there are several warning flags in New York. The challenge for our state has been to manage the increase and try to ensure the spikes in other states don't impact us too much," Governor Cuomo said. "We manage the increase by deploying the most aggressive testing in the country and our micro-cluster strategy - when we identify a small spread, we increase enforcement and restrictions in a targeted way. As we head into winter months, it's going to take the work of all New Yorkers to ensure we don't go back to where we were this spring - I understand COVID fatigue is real, but it's on all of us to stay vigilant and wear our masks, wash our hands, remain socially distant and stay New York Tough."
The Governor noted that the positive testing rate in all focus areas under the state's Micro-Cluster strategy is 4.22 percent, and outside the focus zone areas is 2.01 percent. Within the focus areas, 13,488 test results were reported yesterday, yielding 569 positives. In the remainder of the state, not counting these focus areas, 149,803 test results were reported, yielding 3,018 positives.
The vaccine announcement was widely greeted as a sorely needed positive development in the fight against the virus, which is raging across the country.
Here are a few of the main points about the vaccine:
“Today is a great day for science and humanity. The first set of results from our Phase 3 COVID-19 vaccine trial provides the initial evidence of our vaccine’s ability to prevent COVID-19,” said Dr. Albert Bourla, Pfizer Chairman and CEO. “We are reaching this critical milestone in our vaccine development program at a time when the world needs it most with infection rates setting new records, hospitals nearing over-capacity and economies struggling to reopen. With today’s news, we are a significant step closer to providing people around the world with a much-needed breakthrough to help bring an end to this global health crisis. We look forward to sharing additional efficacy and safety data generated from thousands of participants in the coming weeks.”
After discussion with the FDA, the companies recently elected to drop the 32-case interim analysis and conduct the first interim analysis at a minimum of 62 cases.
Participants in trials will continue to be monitored for long-term protection and safety for an additional two years after their second dose.
Along with the efficacy data generated from the clinical trial, Pfizer and BioNTech are working to prepare the necessary safety and manufacturing data to submit to the FDA to demonstrate the safety and quality of the vaccine product produced.
Based on current projections they expect to produce globally up to 50 million vaccine doses in 2020 and up to 1.3 billion doses in 2021.
While high-risk groups like health care workers and the elderly could get the vaccine later this year, many experts say the general public is not expected to get a vaccine until sometime several months into 2021.
Secretary of Health and Human Services Alex Azar predicted Tuesday that there would be enough Covid-19 vaccine for general public vaccination campaigns by spring 2021.
FDA U.S. health officials have allowed emergency use of the first antibody drug to help the immune system fight COVID-19, an experimental approach against the virus that has killed more than 238,000 Americans. The Food and Drug Administration on Monday cleared the experimental drug from Eli Lilly for people 12 and older with mild or moderate COVID-19 not requiring hospitalization. It's a one-time treatment given through an IV.
The therapy is still undergoing additional testing to establish its safety and effectiveness. It is similar to a treatment President Donald Trump received after contracting the virus last month.
Early results suggest the drug, called bamlanivimab, may help clear the coronavirus sooner and possibly cut hospitalizations in people with mild to moderate COVID-19. A study of it in hospitalized patients was stopped when independent monitors saw the drug did not seem to be helping in that situation.
The government previously reached an agreement to buy and supply much of the early production of Lilly's drug.
Only one drug -- Gilead Sciences’ remdesivir -- has full FDA approval for treating COVID-19. Government treatment guidelines also back using dexamethasone and other steroids for certain severely ill, hospitalized patients.
One other treatment has an emergency use designation now — convalescent plasma, or the blood of COVID-19 survivors. No large studies have shown it to be more effective than usual care alone, however.
The new drug is part of an emerging family of biologic therapies that offer a promising new approach to preventing serious disease and death from COVID-19. Experts say the infused drugs could serve as a therapeutic bridge to help manage the virus until vaccines are widely available.
The drugs are laboratory-made versions of antibodies, blood proteins which the body creates to help target and eliminate foreign infections. The new therapies are concentrated versions of the antibodies that proved most effective against the virus in patient studies.
Regeneron Pharmaceuticals Inc. also has asked for emergency authorization for an antibody drug it is testing, the one Trump received.
FDA regulators authorized the Lilly drug using their emergency powers to quickly speed the availability of experimental drugs and other medical products during public health crises.
In normal times the FDA requires “substantial evidence” to show that a drug is safe and effective, usually through one or more large, rigorously controlled patient studies. But during public health emergencies the agency can lower those standards and require only that an experimental treatment’s potential benefits outweigh its risks.
The emergency authorization functions like a temporary approval for the duration of the COVID-19 pandemic. To win full approval, Lilly will have to submit additional research to fully define the drug’s safety and benefit for patients.
The government has signed an agreement with Lilly to spend $375 million to buy 300,000 vials of the drug. How many doses that would provide is unclear. Each vial contains 700 milligrams and that dose proved ineffective in the early results. It took four times that amount — 2,800 milligrams — to show any effect.
The Lilly drug is authorized for people 12 and older who weigh at least 40 kilograms (about 88 pounds), and who are at high risk for progressing to severe COVID-19 and/or hospitalization. This includes those who are 65 years of age or older, or who have certain chronic medical conditions.
___
AP chief medical writer Marilynn Marchione in Milwaukee contributed to this report.